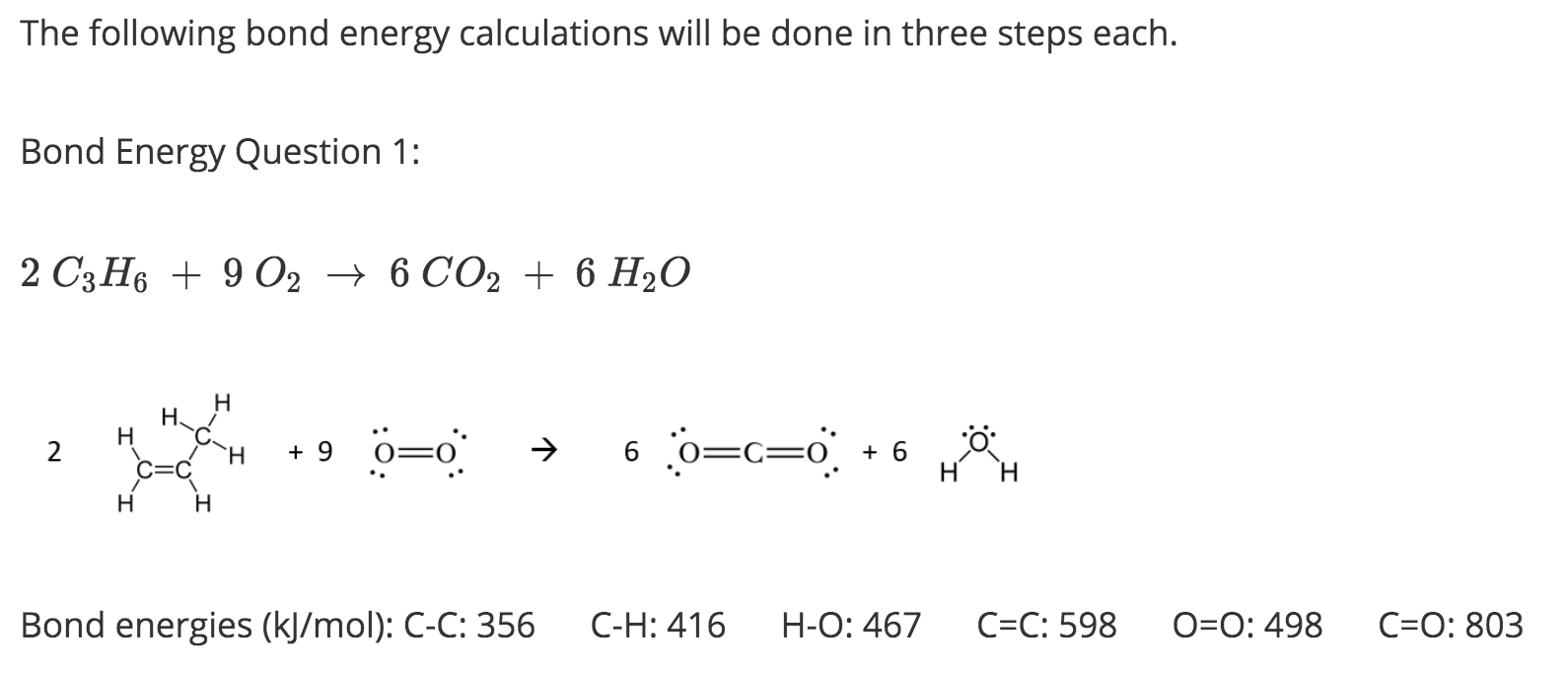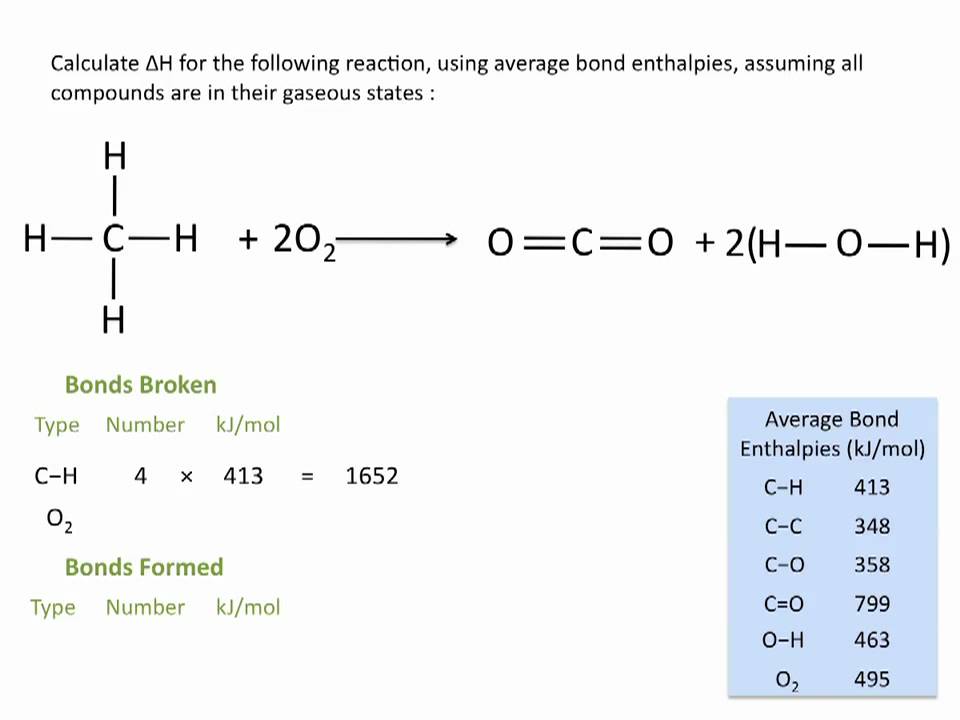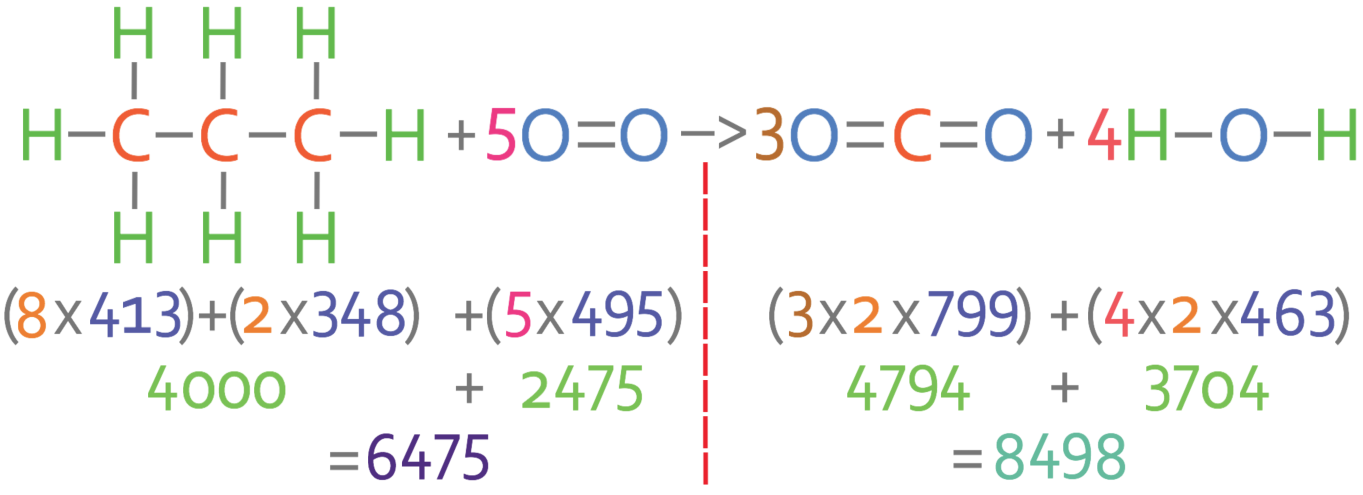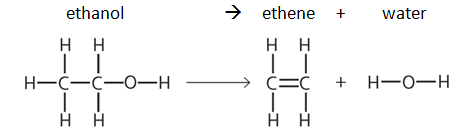
3:07 (Triple only) use bond energies to calculate the enthalpy change during a chemical reaction - TutorMyself Chemistry

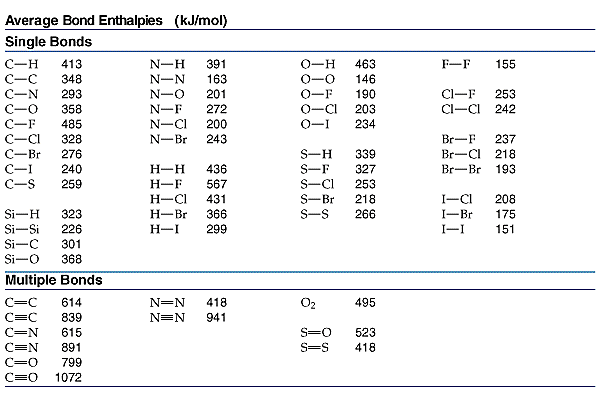
Use the bond energies to estimate Δ H for this reaction: H2(g) + O2(g)⟶H2O2(g) BondBond energy H - H 436 kJ mol^-1 O - O 142 kJ mol^-1 O = O 499